16 grams of helium gas (He) applies 380 mmHg pressure at a temperature of 0 °C. Assume ideal gas behavior. - brainly.com
What is the pressure (atm) of 3.5 moles of helium at -50 °C in a rigid container whose volume is 25.0 L? | Socratic
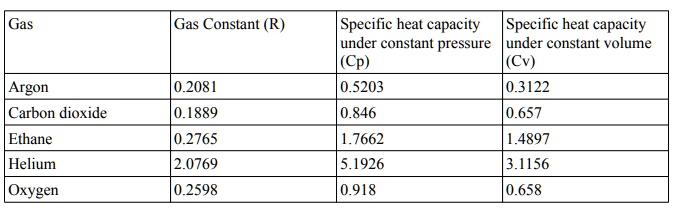
SOLVED: Gas Gas Constant (R) Specilic heat capacity Specilic heat capacity under constant pressure under constant volume Cp) (Cv) 0.5203 0.3122 Argon Carbon dioxide 0.2081 0.1889 0.2765 2.0769 0.2598 0.846 0.657 Ethane 1.7662 5.1926 1.4897 3.1156 ...

The picture (not to scale) shows a pV diagram for 3.6 g of a helium gas (He) that undergoes the process 1 to 2 to 3 to 1. The ideal gas constant
Half mole of Helium gas is contained in a container at STP. Heat energy needed to double pressure of a gas , keeping the volume constant(heat capacity of gas =3j/gm K^°) 1)3276j

OneClass: The molecular mass of helium is 4 g/mol, the Boltzmann's constant is 1.38066 × 10-23 J/K, t...

Hot Selling Helium Balloon Gas 99.999% Helium Gas Cheap Price - China Helium Gas Constant, Helium Gas Cost | Made-in-China.com
![Two moles of Helium gas are to be taken over the cycle \\[{\\text{ABCDA}}\\]as shown (below) in the \\[{\\text{P - T}}\\]diagram. (Assume the gas to be ideal and R is a gas constant.).Now, Two moles of Helium gas are to be taken over the cycle \\[{\\text{ABCDA}}\\]as shown (below) in the \\[{\\text{P - T}}\\]diagram. (Assume the gas to be ideal and R is a gas constant.).Now,](https://www.vedantu.com/question-sets/0cb3c7f1-24ca-4773-848c-92578a8aa4e55598971945812544228.png)